Julian Stubbs has been awarded the Lifetime Achievement Award by SAMPS...
Silk Road Medical
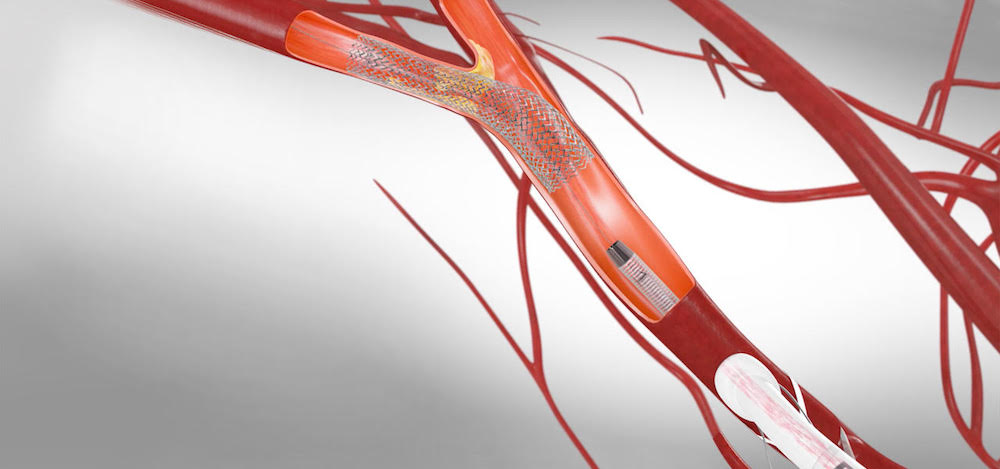
UP developed a campaign, website and communication strategy to showcase how a Silk Road Medical innovation allows vascular surgeons to address carotid artery disease while reducing the risk of stroke and side effects.
View case study